Waec Chemistry practical answers 2022
Here is the Waec Chemistry practical answers 2022
Now its unfair for some of you not to subscribe to our YouTube channel.
For this answers to drop, you need to click on the video below, watch some videoes on our YouTube and then subscribe. We lost alot of subscription already.
https://youtu.be/baINMVXHluE
After watching the video, subscribe here
https://www.youtube.com/channel/UC3QGRuCTKR_YCu10BDrxbyw
and come back here for chemistry practical answers. No watching and subscription, no answer.
Chemistry practical answers
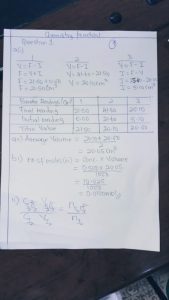
(1a)
Volume of pipette used, VA = 25.0cm³
[TABULATE]
Burette Reading | Rough titre | 1st titre | 2nd titre |
Final Burette reading (cm³) | 25.60 | 32.80 | 26.40
Initial Burette reading (cm³) | 1.20 | 8.70 | 2.30
Volume of Na₂S₂O₃ used | 24.40 | 24.10 | 24.10
Average burette reading = (24.10 + 24.10)/2
Average volume of Na₂S₂O₃ used = 24.10cm³
VB = 24.10cm³
1bi)
Given: CB = 0.1mol/dm³
VB = 24.10cm³
VA = 25.0cm³
CA =?
From equation nA/nB = 1/2
.: Using (CABA)/CBVB = (nA)/nB
(CA x 25)/(0.1×24.10) = 1/2
CA = (0.1×24.10)/(25×2)
CA = 0.0482mol/dm³
.: Concentration of iodine in A = 0.0482mol/dm³
(1bii)
Mass in gramms of iodine in 1dm³ of A = Molarity × Molar mass of iodine
= 0.0482×127(2)
= 12.2428g/dm³
=============================
(2a)
[TABULATE]
=TEST=
C + Distilled water
=OBSERVATION=
It dissolves completely to give a light green solution.
=INFERENCE=
Soluble salt.
(2ai)
=TEST=
Solution C + NaOH in drops and in excess + Heat gently
=OBSERVATION=
A dirty green precipitate is formed which remains insoluble in excess.
Effervescence occurs in which a colourless gas with a pungent smell which turns red litmus blue is given off.
=INFERENCE=
Fe²⁺ is present.
NH₃ gas form.
NH₄⁺ is present.
(2aii)
=TEST=
Solution + BaCl₂ + dilute HCL in excess
=OBSERVATION=
A white precipitate is formed.
The white precipitate remains insoluble and gives a white dense forms
=INFERENCE=
SO₄²⁻, CO₃²⁻, SO₃²⁻, is present.
SO₄²⁻ confirmed.
(2b)
Cations → Fe²⁺ and NH₄⁺
Anions → SO₄²⁻
=============================
(3a)
Carbon (iv) oxide turns like water milky white while sulphur (iv) oxide does not
(3b)
(I) H₂: Downward displacement of air
(II) NH₃: Downward displacement of air
(III) HCL: Upward displacement of air
(3bii)
I – It is less dense than air
II – It is less dense than air
III – It is denser than air
(3c)
(i) Distillation
(ii) Filtration followed by evaporation to dryness
(3d)
This is because KCl react with NaHCO₃ to form two salts
=============================
Completed
(3c)
(i) Fractional distillation
(ii) Crystalization
(3a)
(3bi)
(I) H2 ; downward displacement of water
(II) NH3; downward displacement of air
(III) HCL; upward displacement of air
(3bii)
(I) Hydrogen gas(H2) is collected by the downward displacement of water because It is insoluble in water and It form an explosive mixture with air.
(II) Ammonia gas(NH3) is collected by downward displacement of air because it is lighter than air
(III) HCl gas is collected by upward displacement of air because it is 1.28 times heavier than air.
(3c)
(i) Distillation
(ii) Filtration followed by evaporation to dryness
(3d)
More coming







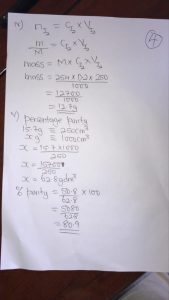

(4a) Molar Concentration of Na²CO³ Solution=0.1mol/dm³
Molar mass of Na²CO³=106gg/mol
Mass Concentration= Molar Concentration× Molar mass
0.1mol/dm³× 106g/mol=10.6g/dm³
This means 1000cm³ of 0.1Mol contains 10.6g of Na²CO³.
:• 300×10.6/1000=3.18g of Na²CO³
We always gat you all
